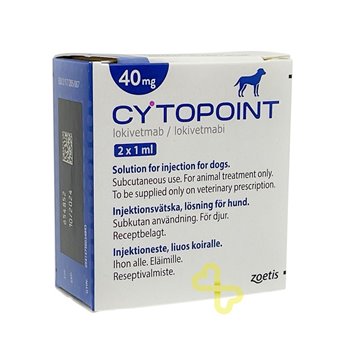
Cytopoint 30mg - Pack of 2 Vials
902883
30mg Cytopoint for Dogs is a prescription injection used to provide relief to dogs suffering from atopic dermatitis, a common and often debilitating skin condition. Cytopoint helps reduce the itchiness and inflammation associated with atopic dermatitis, allowing dogs to live more comfortably. Pack of 2 Vials.
30mg Cytopoint is a refrigerated medication - We will send it via Royal Mail Special Delivery using insulated packaging and ice packs. A signature is required on delivery and it must be put straight into a fridge. We cannot guarantee same day dispatch for orders placed after 12pm. VETERINARY PRESCRIPTION REQUIRED.
Atopic dermatitis is a chronic inflammatory skin disease caused by allergic reactions to environmental substances such as pollen, moulds, dust mites, and certain foods. Symptoms typically include severe itching, redness, and recurrent skin infections. This condition can significantly impact a dog's quality of life, leading to discomfort, anxiety, and behavioural changes.
Dogs with atopic dermatitis often exhibit excessive scratching, licking, chewing, or rubbing of their skin. These behaviours can lead to hair loss, open sores, and secondary bacterial or yeast infections. Commonly affected areas include the face, ears, paws, abdomen, and armpits. If left untreated, the condition can worsen, causing chronic skin damage and increased susceptibility to infections.
Cytopoint 30mg for Dogs is a biological therapy that works by mimicking the natural immune system response. It contains a monoclonal antibody specifically designed to bind to canine IL-31. When injected, these antibodies circulate in the dog's body and neutralise IL-31, effectively blocking the itch signal. This action provides rapid relief, often within 24 hours, and can last for up to four to eight weeks, depending on the individual dog's response. This duration of action allows for less frequent dosing compared to traditional therapies such as tablets, making it a convenient option.
Product Features
- Pack Size: - Pack of 2 Vials
- Target Animal: - Dog (Canine)
- Related Condition: - Atopic Dermatitis in Dogs
- Pet Prescription Required?: - Yes
- Active Ingredient: - Monoclonal antibody
- Product Name: - 30mg Cytopoint for Dogs
More Information
Description
Cytopoint contains lokivetmab, a caninised monoclonal antibody. It is a targeted biological therapy designed to recognise and neutralise a key itch messenger called interleukin-31, often shortened to IL-31. IL-31 plays an important role in triggering pruritus, the clinical term for itching, in many dogs with allergic skin disease. By binding to IL-31, 30mg Cytopoint helps interrupt the signal that tells your dog’s body to itch.
Once a dog starts scratching, chewing paws or rubbing the face and ears, the skin barrier can become more damaged and inflamed. 30mg Cytopoint helps break that itch cycle in a focused way. 30mg Cytopoint is not a steroid and is not used in the same way as broad immunosuppressive medication. It is normally given by injection by a veterinary professional or at home after training, and your vet will decide whether 30mg Cytopoint is the right strength for your dog.
Atopic dermatitis in dogs is a chronic inflammatory skin disease associated with allergic hypersensitivity, most often to environmental allergens such as pollen, house dust mites, moulds and similar triggers. Dogs with atopic dermatitis often have an impaired skin barrier, which means the skin is less effective at protecting itself from irritation and allergens. This can allow inflammation to develop more easily and can make flare-ups more common. Although every dog is different, common signs can include:
- Persistent scratching or rubbing
- Licking or chewing at the paws
- Recurrent ear irritation or otitis externa
- Red, inflamed or sensitive skin
- Hair thinning or hair loss
- Skin darkening or thickening over time
- Recurrent bacterial or yeast skin infections
- Face rubbing, scooting or restless behaviour
Atopic dermatitis is not always obvious in its early stages. Some dogs begin with mild paw licking or ear irritation before more widespread skin changes appear. Others may only seem itchy seasonally at first, then become uncomfortable for longer periods. Because allergic skin disease is often multifactorial, your vet may also consider other issues such as flea allergy dermatitis, food reactions, pyoderma or Malassezia overgrowth when deciding whether 30mg Cytopoint is appropriate. In that setting, 30mg Cytopoint can be particularly helpful because reducing pruritus may give the skin a better chance to recover while any secondary problems are identified and treated.
For dogs affected by allergic dermatitis, Cytopoint can be a very useful option because it focuses on relieving itch without relying on a blanket approach. That can be especially valuable for dogs who have repeated flare-ups, dogs who are damaging their own skin by scratching, or dogs whose owners are looking for a more targeted way to control discomfort. When a dog is less itchy, there is often less licking, less chewing at the paws, less rubbing around the eyes and muzzle, and fewer opportunities for the skin to become traumatised. In some cases, that may also reduce the chance of secondary skin infection developing or recurring.
It is also important to understand that 30mg Cytopoint helps manage the itch associated with allergic skin disease, but it does not remove the underlying allergic tendency. Dogs with atopic dermatitis often need broader skin support as well. Depending on the case, that may include medicated or soothing shampoos, skin barrier products, regular parasite control, ear cleaners, topical sprays, essential fatty acid support, or treatment for secondary bacterial or yeast infection. Some dogs may also need further investigation into their triggers, particularly if flare-ups are frequent or severe. For this reason, 30mg Cytopoint often works best as part of a wider dermatology plan rather than in isolation. Owners sometimes worry if a dog still needs bathing, ear care or topical support after Cytopoint, but this is very common in canine dermatology. 30mg Cytopoint can reduce the urge to scratch, while the rest of the treatment plan supports the skin itself. That combination can be much more effective than trying to rely on one product alone.
What is 30mg Cytopoint used for in dogs?
30mg Cytopoint is used to help control allergic itching in dogs, particularly itch linked to atopic dermatitis. It is prescribed by a vet as part of an individual treatment plan and is intended to reduce the urge to scratch, chew, lick and rub irritated skin.
How does 30mg Cytopoint work?
30mg Cytopoint works by targeting interleukin-31, a protein involved in sending itch signals in allergic dogs. It contains lokivetmab, a monoclonal antibody that binds to this itch messenger, helping reduce pruritus in a focused way rather than using a broad anti-inflammatory effect.
Is Cytopoint a steroid?
No, 30mg Cytopoint is not a steroid. It is a monoclonal antibody treatment designed to target one of the key itch pathways in dogs. Many owners and vets value this because it offers a different clinical approach to managing allergic itch.
How quickly can 30mg Cytopoint start working?
Many dogs show improvement fairly quickly after receiving Cytopoint, although the exact response time can vary. Some become less itchy within a short period, while others improve more gradually as the skin begins to settle and the itch-scratch cycle starts to break.
How long does Cytopoint last?
The effect of 30mg Cytopoint can vary between dogs, which is why your vet will advise on timing for repeat treatment. Some dogs remain comfortable for several weeks, while others may need closer monitoring and a dosing schedule that reflects their own pattern of allergic flare-ups.